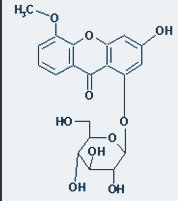
Xanthone und ihre Bedeutung
Mangostan enthält über 40 Xanthone und ist damit die reichhaltigste Quelle natürlicher Xanthone. Keine andere Frucht kann das leisten. Durchgeführter klinischer Studien und Laboruntersuchungen haben gezeigt, dass die Xanthone in der Mangostan für eine Vielzahl verschiedener medizinischer Nutzen verantwortlich sind.
Folgende nützliche Wirkungen wurden bis jetzt wissenschaftlich untersucht und bestätigt (Studien):
- antientzündlich
- anti-oxidativ
- bakterizid, fungizid, antiviral
- effektiv gegen Krebs und sonstige Geschwüre
- anti-hepatotoxisch
 anti-allergisch
anti-allergisch
Wissenschaftlichen Untersuchungen in Thailand, Sri Lanka und Japan zufolge werden die Xanthone der Mangostan in Zukunft einen wertvollen Beitrag bei der Prävention und der Behandlung von Krebserkrankungen spielen. Zudem wirkt Gamma-Mangostan schmerzlindernd und entzündungshemmend. Es hemmt die Cyclooxygenase (COX-1 und COX-2) ohne schädliche Nebenwirkung.
XANTHONE – Aktuelle wissenschaftliche Studien und Forschungsergebnisse
Quelle: Datenbank der U.S. National Library of Medicine and the National Institutes of Health (siehe: http://www.ncbi.nlm.nih.gov/sites/entrez)
1: Phytochemistry. 2007 Nov 6
Xanthones with quinone reductase-inducing activity from the fruits of Garcinia mangostana (Mangosteen).
Chin YW, Jung HA, Chai H, Keller WJ, Kinghorn AD.
Division of Medicinal Chemistry and Pharmacognosy, College of Pharmacy, The Ohio State University, Columbus, OH 43210, USA.
Bioactivity-guided fractionation of a dichloromethane-soluble extract of Garcinia mangostana fruits has led to the isolation and identification of five compounds, including two xanthones, 1,2-dihydro-1, 8, 10-trihydroxy-2- (2-hydroxypropan-2-yl) -9-(3-methylbut-2-enyl) furo[3,2-a] xanthen-11-one (1) and 6-deoxy-7-demethylmangostanin (2), along with three known compounds, 1,3,7-trihydroxy-2,8-di-(3-methylbut-2-enyl)xanthone (3), mangostanin (4), and alpha-mangostin (5). The structures of compounds 1 and 2 were determined from analysis of their spectroscopic data. All isolated compounds in the present study together with eleven other compounds previously isolated from the pericarp of mangosteen, were tested in an in vitro quinone reductase-induction assay using murine hepatoma cells (Hepa 1c1c7) and an in vitro hydroxyl radical antioxidant assay. Of these, compounds 1-4 induced quinone reductase (concentration to double enzyme induction, 0.68-2.2mug/mL) in Hepa 1c1c7 cells and gamma-mangostin (6) exhibited hydroxyl radical-scavenging activity (IC(50), 0.20mug/mL).
PMID: 17991497 [PubMed - as supplied by publisher]
2: J Agric Food Chem. 2007 Nov 28;55(24):9805-10. Epub 2007 Oct 26.
Assay-guided Fractionation Study of alpha-Amylase Inhibitors from Garcinia mangostana Pericarp.
Eng Kiat Loo A, Huang D.
chmhdj@nus.edu.sg.
alpha-Amylase inhibitor (alpha-AI) activity of Garcinia mangostana, commonly known as mangosteen, pericarp extracts was studied by assay guided fractionations from lipophilic to hydrophilic using combined solvent extraction and Amberlite XAD2 adsorption chromatography. Neither the lipophilic, xanthone containing fraction, nor the highly polar fraction, which has no affinity on Amberlite XAD2, showed any alpha-AI. The fraction that shows very high inhibitory activity contains primarily polyphenols and can be adsorbed on Amberlite XAD2. The IC50 of 5.4 microg/mL of this fraction is comparable to that of acarbose, a prescribed alpha-AI used in the control of type II diabetes, at 5.2 microg/mL. Total phenolic content (TPC) of each fraction was measured and the TPC has no correlation with the alpha-AI activity. The lipophilic fraction contains mainly xanthones as revealed by HPLC-MS analysis. Colorimetric analysis coupled with UV-vis and IR spectroscopic analysis demonstrated that the fractions with high alpha-AI activity are primarily oligomeric proanthocyanidins (OPCs) with little gallate moiety. There is also evidence to show that the alpha-AI by these OPCs is not purely by nonspecific protein complexation. Both tannic acid and G. mangostana OPCs precipitate BSA equally well but G. mangostana OPCs are 56 times more effective in inhibiting alpha-amylase.
PMID: 17960880 [PubMed - in process]
3: Bioorg Med Chem. 2007 Aug 15;15(16):5620-8. Epub 2007 May 18.
Characterized mechanism of alpha-mangostin-induced cell death: caspase-independent apoptosis with release of endonuclease-G from mitochondria and increased miR-143 expression in human colorectal cancer DLD-1 cells.
Nakagawa Y, Iinuma M, Naoe T, Nozawa Y, Akao Y.
Gifu International Institute of Biotechnology, 1-1 Naka-Fudogaoka, Kakamigahara, Gifu 504-0838, Japan.
alpha-Mangostin, a xanthone from the pericarps of mangosteen (Garcinia mangostana Linn.), was evaluated for in vitro cytotoxicity against human colon cancer DLD-1 cells. The number of viable cells was consistently decreased by the treatment with alpha-mangostin at more than 20 microM. The cytotoxic effect of 20 microM alpha-mangostin was found to be mainly due to apoptosis, as indicated by morphological findings. Western blotting, the results of an apoptosis inhibition assay using caspase inhibitors, and the examination of caspase activity did not demonstrate the activation of any of the caspases tested. However, endonuclease-G released from mitochondria with the decreased mitochondrial membrane potential was shown. The levels of phospho-Erk1/2 were increased in the early phase until 1h after the start of treatment and thereafter decreased, and increased again in the late phase. On the other hand, the level of phospho-Akt was sharply reduced with the process of apoptosis after 6h of treatment. Interestingly, the level of microRNA-143, which negatively regulates Erk5 at translation, gradually increased until 24h following the start of treatment. We also examined the synergistic growth suppression in DLD-1 cells by the combined treatment of the cells with alpha-mangostin and 5-FU which is one of the most effective chemotherapeutic agents for colorectal adenocarcinoma. The co-treatment with alpha-mangostin and 5-FU, both at 2.5 microM, augmented growth inhibition compared with the treatment with 5 microM of alpha-mangostin or 5 microM 5-FU alone. These findings indicate unique mechanisms of alpha-mangostin-induced apoptosis and its action as an effective chemosensitizer.
Publication Types:
PMID: 17553685 [PubMed - indexed for MEDLINE]
4: J Pharm Biomed Anal. 2007 Mar 12;43(4):1270-6. Epub 2006 Nov 28.
Quantitative and qualitative determination of six xanthones in Garcinia mangostana L. by LC-PDA and LC-ESI-MS.
Ji X, Avula B, Khan IA.
National Center for Natural Products Research, Research Institute of Pharmaceutical Sciences, School of Pharmacy, The University of Mississippi, University, MS 38677, USA.
A new method was developed for the simultaneous analysis of six naturally occurring xanthones (3-isomangostin, 8-desoxygartanin, gartanin, alpha-mangostin, 9-hydroxycalabaxanthone and beta-mangostin). The quantitative determination was conducted by reversed phase high performance liquid chromatography with photodiode array detector (LC-PDA). Separation was performed on a Phenomenex Luna C18(2) (150 mm x 3.00 mm, 5 microm) column. The xanthones were identified by retention time, ultraviolet (UV) spectra and quantified by LC-PDA at 320 nm. The precision of the method was confirmed by the relative standard deviation (R.S.D.), which was < or =4.6%. The recovery was in the range from 96.58% to 113.45%. A good linear relationship was established in over two orders of magnitude range. The limits of detection (LOD) for six xanthone compounds were < or =0.248 microg/mL. The identity of the peaks was further confirmed by high performance liquid chromatography with time-of-flight mass spectrometry (LC-TOF MS) system coupled with electrospray ionization (ESI) interface. The developed methods were applied to the determination of six xanthones in Garcinia mangostana products. The satisfactory results showed that the methods are effective for the analysis of real samples.
Publication Types:
PMID: 17129697 [PubMed - indexed for MEDLINE]
5: Nat Prod Res. 2006 Oct;20(12):1067-73.
Xanthones from Garcinia mangostana (Guttiferae).
Ee GC, Daud S, Taufiq-Yap YH, Ismail NH, Rahmani M.
Department of Chemistry, Faculty of Science, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia. gwen@fsas.upm.edu.my
Studies on the stem of Garcinia mangostana have led to the isolation of one new xanthone mangosharin (1) (2,6-dihydroxy-8-methoxy-5-(3-methylbut-2-enyl)-xanthone) and six other prenylated xanthones, alpha-mangostin (2), beta-mangostin (3), garcinone D (4), 1,6-dihydroxy-3,7-dimethoxy-2-(3-methylbut-2-enyl)-xanthone (5), mangostanol (6) and 5,9-dihydroxy-8- methoxy-2,2-dimethyl-7-(3-methylbut-2-enyl)-2H,6H-pyrano-[3,2-b]-xanthene-6-one (7). The structures of these compounds were determined by spectroscopic methods such as 1H NMR, 13C NMR, mass spectrometry (MS) and by comparison with previous studies. All the crude extracts when screened for their larvicidal activities indicated very good toxicity against the larvae of Aedes aegypti. This article reports the isolation and identification of the above compounds as well as bioassay data for the crude extracts. These bioassay data have not been reported before.
Publication Types:
PMID: 17127660 [PubMed - indexed for MEDLINE]
6: Planta Med. 2006 Aug;72(10):912-6. Epub 2006 Aug 10.
Prenylated xanthones as potential antiplasmodial substances.
Mahabusarakam W, Kuaha K, Wilairat P, Taylor WC.
Department of Chemistry, Prince of Songkla University, Hat Yai, Songkhla, Thailand. wilawan.m@psu.ac.th
Mangostin, the major xanthone of Garcinia mangostana, and a series of synthetic derivatives were investigated for their in vitro antiplasmodial activity against Plasmodium falciparum. Mangostin itself showed moderate activity, but prenylated xanthones containing alkylamino functional groups exhibited quite potent antiplasmodial activity. Some structure-activity relationships are proposed.
Publication Types:
PMID: 16902859 [PubMed - indexed for MEDLINE]
7: Phytochemistry. 2005 Jul;66(14):1718-23.
Xanthones and benzophenones from Garcinia griffithii and Garcinia mangostana.
Nguyen LH, Venkatraman G, Sim KY, Harrison LJ.
Department of Chemistry, National University of Singapore, 3 Science Drive 3, Singapore 117543, Republic of Singapore.
A new polyisoprenylated benzophenone, guttiferone I, together with the known compounds cambogin, 1,7-dihydroxyxanthone, 1,3,6,7-tetrahydroxyxanthone and 1,3,5,6-tetrahydroxyxanthone were isolated from the stem bark of Garcinia griffithii. The acetone extract of the heartwood of Garcinia mangostana contained one new diprenylated xanthone (mangoxanthone) and a new benzophenone (3',6-dihydroxy-2,4,4'-trimethoxybenzophenone) as well as the known xanthones dulxanthone D, 1,3,7-trihydroxy-2-methoxyxanthone, 1,3,5-trihydroxy-13,13-dimethyl-2H-pyran[7,6-b]xanthen-9-one. Their structures were established on the basis of spectroscopic studies and chemical correlation.
Publication Types:
PMID: 16173114 [PubMed - indexed for MEDLINE]
8: J Ethnopharmacol. 2005 Oct 3;101(1-3):330-3.
Antimicrobial effects of Thai medicinal plants against acne-inducing bacteria.
Chomnawang MT, Surassmo S, Nukoolkarn VS, Gritsanapan W.
Department of Microbiology, Faculty of Pharmacy, Mahidol University, 447 Sri Ayudthaya Road, Rachathevi, Bangkok 10400, Thailand. scmtd@mahidol.ac.th
Propionibacterium acnes and Staphylococcus epidermidis have been recognized as pus-forming bacteria triggering an inflammation in acne. The present study was conducted to evaluate antimicrobial activities of Thai medicinal plants against these etiologic agents of acne vulgaris. Crude extracts were tested for antimicrobial activities by disc diffusion and broth dilution methods. The results from the disc diffusion method showed that 13 medicinal plants could inhibit the growth of Propionibacterium acnes. Among those, Senna alata, Eupatorium odoratum, Garcinia mangostana, and Barleria lupulina had strong inhibitory effects. Based on a broth dilution method, the Garcinia mangostana extract had the greatest antimicrobial effect. The MIC values were the same (0.039 mg/ml) for both bacterial species and the MBC values were 0.039 and 0.156 mg/ml against Propionibacterium acnes and Staphylococcus epidermidis, respectively. In bioautography assay, the Garcinia mangostana extract produced strong inhibition zones against Propionibacterium acnes. Antimicrobial activity from fractions of column chromatography revealed one of the active compounds in Garcinia mangostana could be mangostin, a xanthone derivative. Taken together, our data indicated that Garcinia mangostana had a strong inhibitory effect on Propionibacterium acnes and Staphylococcus epidermidis. Therefore, this plant would be an interesting topic for further study and possibly for an alternative treatment for acne.
Publication Types:
PMID: 16009519 [PubMed - indexed for MEDLINE]
9: Asian Pac J Cancer Prev. 2004 Oct-Dec;5(4):433-8.
Inhibitory effects of crude alpha-mangostin, a xanthone derivative, on two different categories of colon preneoplastic lesions induced by 1, 2-dimethylhydrazine in the rat.
Nabandith V, Suzui M, Morioka T, Kaneshiro T, Kinjo T, Matsumoto K, Akao Y, Iinuma M, Yoshimi N.
Tumor Pathology Division, Faculty of Medicine, University of the Ryukyus, Okinawa 903-0215, Japan.
The purpose of this study was to examine whether crude alpha-mangostin (a major xanthone derivative in mangosteen pericarp (Garcinia mangostana)) has short-term chemopreventive effects on putative preneoplastic lesions involved in rat colon carcinogenesis. The crude preparation was obtained by simple recrystallization of an ethylacetate extract of mangosteen pericarps. A total of 33 five-week-old male F344 rats were randomly divided into 5 experimental groups. Rats in groups 1-3 were given a subcutaneous injection of 1,2-dimethylhydrazine (DMH)(40 mg/kg body weight) once a week for 2 weeks. Starting one week before the first injection of DMH, rats in groups 2 and 3 were fed a diet containing 0.02% and 0.05% crude alpha-mangostin, respectively, for 5 weeks. Rats in group 4 also received the diet containing 0.05% crude alpha-mangostin, while rats in group 5 served as untreated controls. The experiment was terminated 5 weeks after the start. Dietary administration of crude alpha-mangostin at both doses significantly inhibited the induction and/or development of aberrant crypt foci (ACF) (P<0.05 for 0.02% crude alpha-mangostin, P<0.01 for 0.05% crude alpha-mangostin), when compared to the DMH-treated group (group 1). Moreover, treatment of rats with 0.05% crude alpha-mangostin significantly decreased dysplastic foci (DF) (P<0.05) and beta-catenin accumulated crypts (BCAC) (P<0.05), to below the group 1 values. The proliferating cell nuclear antigen (PCNA) labeling indices of colon epithelium and focal lesions in groups 2 and 3 were also significantly lower than in group 1 and this effect occurred in a dose dependent manner of the crude alpha-mangostin. This finding that crude alpha-mangostin has potent chemopreventive effects in our short-term colon carcinogenesis bioassay system suggests that longer exposure might result in suppression of tumor development.
Publication Types:
PMID: 15546251 [PubMed - indexed for MEDLINE]
10: J Pharmacol Sci. 2004 May;95(1):33-40.
Alpha-mangostin induces Ca2+-ATPase-dependent apoptosis via mitochondrial pathway in PC12 cells.
Sato A, Fujiwara H, Oku H, Ishiguro K, Ohizumi Y.
Department of Pharmaceutical Molecular Biology, Graduate School of Pharmaceutical Sciences, Tohoku University, Sendai, Japan.
We investigated the cell death effects of eight xanthones on PC12 rat pheochromocytoma cells. Among these compounds, alpha-mangostin, from the fruit hull of Garcinia mangostana L., had the most potent effect with the EC(50) value of 4 microM. Alpha-mangostin-treated PC12 cells demonstrated typical apoptotic DNA fragmentation and caspase-3 cleavage (equivalent to activation). The flow cytometric analysis indicated that this compound induced apoptosis in time-and concentration-dependent manners. Alpha-mangostin showed the features of the mitochondrial apoptotic pathway such as mitochondrial membrane depolarization and cytochrome c release. Furthermore, alpha-mangostin inhibited the sarco(endo)plasmic reticulum Ca(2+)-ATPase markedly. There was a correlation between the Ca(2+)-ATPase inhibitory effects and the apoptotic effects of the xanthone derivatives. On the other hand, c-Jun NH(2)-terminal kinase (JNK/SAPK), one of the signaling molecules of endoplasmic reticulum (ER) stress, was activated with alpha-mangostin treatment. These results suggest that alpha-mangostin inhibits Ca(2+)-ATPase to cause apoptosis through the mitochondrial pathway.
Publication Types:
PMID: 15153648 [PubMed - indexed for MEDLINE]
11: Planta Med. 2002 Nov;68(11):975-9.
Garcinone E, a xanthone derivative, has potent cytotoxic effect against hepatocellular carcinoma cell lines.
Ho CK, Huang YL, Chen CC.
Department of Medical Research & Education, Veterans General Hospital, Taipei, ROC.
Treatment of hepatocellular carcinomas (HCCs) with chemotherapy has generally been disappointing and it is most desirable to have more effective new drugs. We extracted and purified 6 xanthone compounds from the rinds (peel) of the fruits of Garcinia mangostana L., using partitioned chromatography and then tested the cytotoxic effects of these compounds on a panel of 14 different human cancer cell lines including 6 hepatoma cell lines, based on the MTT method. Several commonly used chemotherapeutic agents were included in the assay to determine the relative potency of the potential new drugs. Our results have shown that one of the xanthone derivatives which could be identified as garcinone E has potent cytotoxic effect on all HCC cell lines as well as on the other gastric and lung cancer cell lines included in the screen. We suggest that garcinone E may be potentially useful for the treatment of certain types of cancer.
Publication Types:
PMID: 12451486 [PubMed - indexed for MEDLINE]
12: J Nat Prod. 2002 May;65(5):761-3.
Xanthones from the green fruit hulls of Garcinia mangostana.
Suksamrarn S, Suwannapoch N, Ratananukul P, Aroonlerk N, Suksamrarn A.
Department of Chemistry, Faculty of Science, Srinakharinwirot University, Bangkok 10110, Thailand. sunit@psm.swu.ac.th
Three new xanthones, mangostenol (1), mangostenone A (2), and mangostenone B (3), were isolated from the green fruit hulls of Garcinia mangostana, along with the known xanthones, trapezifolixanthone, tovophyllin B (4), alpha- and beta-mangostins, garcinone B, mangostinone, mangostanol, and the flavonoid epicatechin. The structures of the new xanthones were elucidated by analysis of their spectroscopic data.
Publication Types:
PMID: 12027762 [PubMed - indexed for MEDLINE]
13: Biochem Pharmacol. 2002 Jan 1;63(1):73-9.
Inhibition of cyclooxygenase and prostaglandin E2 synthesis by gamma-mangostin, a xanthone derivative in mangosteen, in C6 rat glioma cells.
Nakatani K, Nakahata N, Arakawa T, Yasuda H, Ohizumi Y.
Department of Pharmaceutical Molecular Biology, Graduate School of Pharmaceutical Sciences, Tohoku University, Aoba, Aramaki, Aoba-ku, 980-8578, Sendai, Japan.
The fruit hull of mangosteen, Garcinia mangostana L., has been used for many years as a medicine for treatment of skin infection, wounds, and diarrhea in Southeast Asia. In the present study, we examined the effect of gamma-mangostin, a tetraoxygenated diprenylated xanthone contained in mangosteen, on arachidonic acid (AA) cascade in C6 rat glioma cells. gamma-Mangostin had a potent inhibitory activity of prostaglandin E2 (PGE2) release induced by A23187, a Ca2+ ionophore. The inhibition was concentration-dependent, with the IC50 value of about 5 microM. gamma-Mangostin had no inhibitory effect on A23187-induced phosphorylation of p42/p44 extracellular signal regulated kinase/mitogen-activated protein kinase or on the liberation of [14C]-AA from the cells labeled with [14C]-AA. However, gamma-mangostin concentration-dependently inhibited the conversion of AA to PGE2 in microsomal preparations, showing its possible inhibition of cyclooxygenase (COX). In enzyme assay in vitro, gamma-mangostin inhibited the activities of both constitutive COX (COX-1) and inducible COX (COX-2) in a concentration-dependent manner, with the IC50 values of about 0.8 and 2 microM, respectively. Lineweaver-Burk plot analysis indicated that gamma-mangostin competitively inhibited the activities of both COX-1 and -2. This study is a first demonstration that gamma-mangostin, a xanthone derivative, directly inhibits COX activity.
Publication Types:
PMID: 11754876 [PubMed - indexed for MEDLINE]
14: J Pharm Pharmacol. 1996 Aug;48(8):861-5.
Antibacterial activity of xanthones from guttiferaeous plants against methicillin-resistant Staphylococcus aureus.
Iinuma M, Tosa H, Tanaka T, Asai F, Kobayashi Y, Shimano R, Miyauchi K.
Department of Pharmacognosy, Gifu Pharmaceutical University, Japan.
Extracts of Garcinia mangostana (Guttiferae) showing inhibitory effects against the growth of S. aureus NIHJ 209p were fractionated according to guidance obtained from bioassay and some of the components with activity against methicillin-resistant Staphylococcus aureus (MRSA) were characterized. One active isolate, alpha-mangostin, a xanthone derivative, had a minimum inhibitory concentration (MIC) of 1.57-12.5 micrograms mL-1. Other related xanthones were also examined to determine their anti-MRSA activity. Rubraxanthone, which was isolated from Garcinia dioica and has a structure similar to that of alpha-mangostin, had the highest activity against staphylococcal strains (MIC = 0.31-1.25 micrograms mL-1), an activity which was greater than that of the antibiotic vancomycin (3.13-6.25 micrograms mL-1). The inhibitory effect against strains of MRSA of two of the compounds when used in conjunction with other antibiotics was also studied. The anti-MRSA activity of alpha-mangostin was clearly increased by the presence of vancomycin; this behaviour was not observed for rubraxanthone. The strong in-vitro antibacterial activity of xanthone derivatives against both methicillin-resistant and methicillin-sensitive Staphylococcus aureus suggests the compounds might find wide pharmaceutical use.
Publication Types:
PMID: 8887739 [PubMed - indexed for MEDLINE]
15: Indian J Exp Biol. 1980 Aug;18(8):843-6.
Effect of mangostin, a xanthone from Garcinia mangostana Linn. in immunopathological & inflammatory reactions.
Gopalakrishnan C, Shankaranarayanan D, Kameswaran L, Nazimudeen SK.
PMID: 7461736 [PubMed - indexed for MEDLINE]
16: Arch Int Pharmacodyn Ther. 1979 Jun;239(2):257-69.
Pharmacological profile of mangostin and its derivatives.
Shankaranarayan D, Gopalakrishnan C, Kameswaran L.
Mangostin (M), a naturally occurring xanthone in the rinds of the fruits of Garcinia mangostana Linn. (Guttiferae) and its derivatives such as 3-0-methyl mangostin (MM), 3,6-di-O-methyl mangostin (DM), 1-isomangostin (IM), mangostin triacetate (MT), mangostin 3,6-di-O-(tetra acetyl) glucoside (MTG) and mangostin-6,6-di-O-glucoside (MOG) were screened for various pharmacological effects in experimental animals. With the exception of DM all the test compounds produced CNS depression characterised by ptosis, sedation, decreased motor activity, potentiation of pentobarbital sleeping time and ether anaesthesia in mice and rats. None of the compounds exhibited analgesic, antipyretic and anticonvulsant effects. With the exception of MOG, none of the test compounds produced significant effects on the cardiovascular system of frogs and dogs. MOG produced myocardial stimulation and a rise in blood pressure which was partially blocked by propranolol. M, IM and MT produced pronounced antiinflammatory activity both by intraperitoneal and oral routes in rats as tested by carrageenininduced hind paw oedema, cotton pellet implantation and granuloma pouch techniques. Antiinflammatory activity for M, IM and MT was observed even in bilaterally adrenalectomised rats. M, IM and MT did not produce any mast cell membrane stabilising effect and the degranulation effect of polymyxin B, diazoxide and Triton X-100 on rat peritoneal mast cells in vitro was not prevented. M, IM and MT did not alter the prothrombin time of albino rats. M alone produced significant antiulcer activity in rats.
PMID: 314790 [PubMed - indexed for MEDLINE]




















